Publications
| 2025 (2) | |||
| 2024 (7) | 2023 (5) | 2022 (3) | 2021 (5) |
| 2020 (3) | 2019 (12) | 2018 (5) | 2017 (3) |
| 2016 (4) | 2015 (2) | 2014 (4) | 2013 (7) |
| 2012 (7) | 2011 (1) | 2010 (2) | 2008 (1) |
2019 | Articles and Reviews
[Article] Nanotechnologies : les chimistes au cœur des dernières avancées
|
"Domaine prioritaire dans la R&D des pays industrialisés, les nanotechnologies seront à l’horizon 2030 essentielles aux ingénieurs pour faire face aux crises environnementales, à la raréfaction de matières premières stratégiques et à la demande croissante d’énergie. Les chimistes y jouent un rôle de premier plan." Cet article publié dans le dossier spécial sur la chimie de La Jaune et la Rouge discute du rôle des chimistes dans les nanotechnologies. Sommaire du dossier | Lire l'article. |
 |
|
S. Carenco, La Jaune et la Rouge 2019, 749, 46-47. |
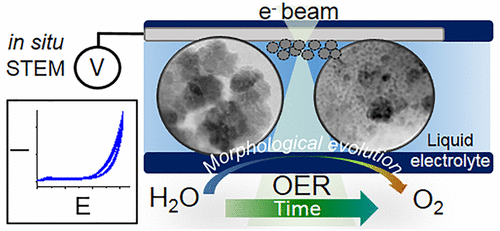
Morphological and Structural Evolution of Co3O4 Nanoparticles Revealed by in Situ Electrochemical Transmission Electron Microscopy during Electrocatalytic Water Oxidation
|
N. Ortiz Peña, D. Ihiawakrim, M. Han, B. Lassalle-Kaiser, S. Carenco, C. Sanchez, C. Laberty-Robert, D. Portehault, O. Ersen, ACS Nano 2019, 13, 11372. |
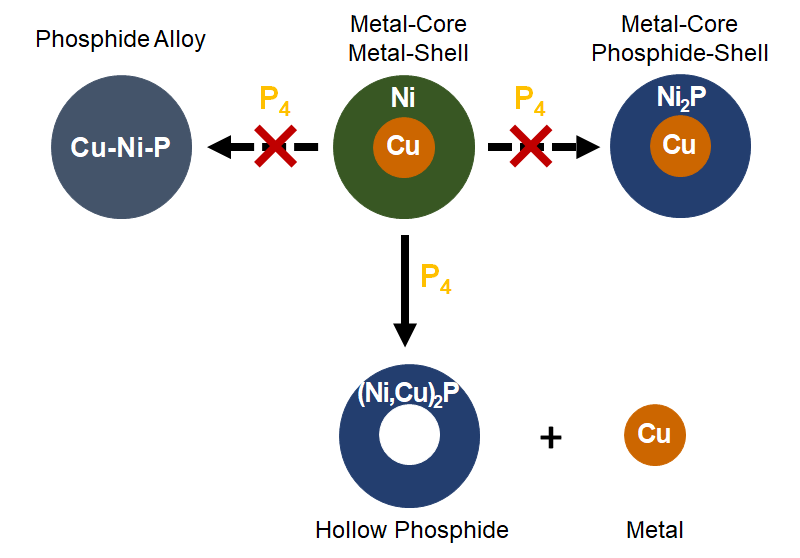
Bimetallic Phosphide (Ni,Cu)2P Nanoparticles by Inward Phosphorus Migration and Outward Copper Migration
|
A.-M. Nguyen, M. Bahri, S. Dreyfuss, S. Moldovan, A. Miche, C. Méthivier, O. Ersen, N. Mézailles, S. Carenco, Chem. Mater. 2019, 31, 6124-34. |
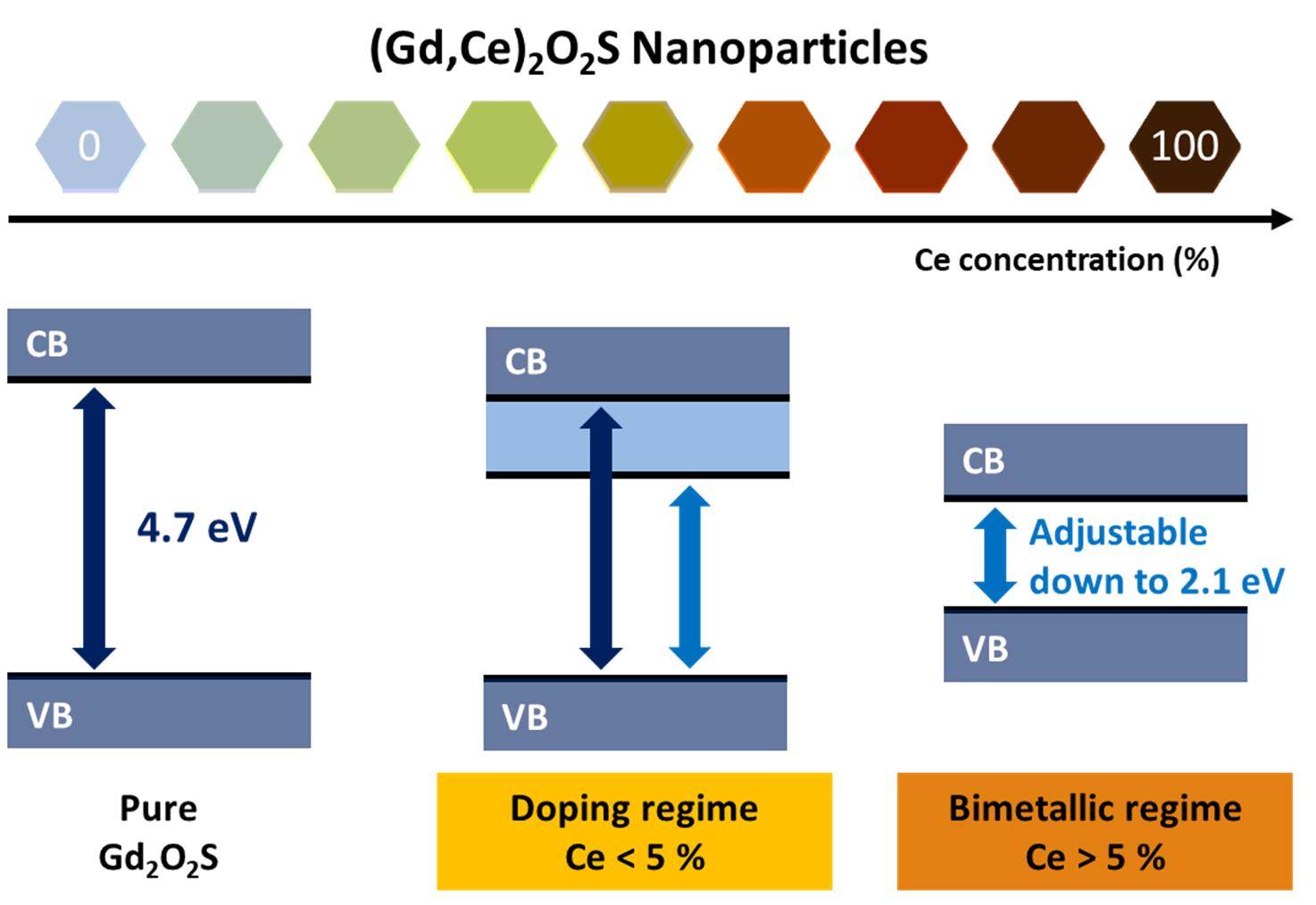
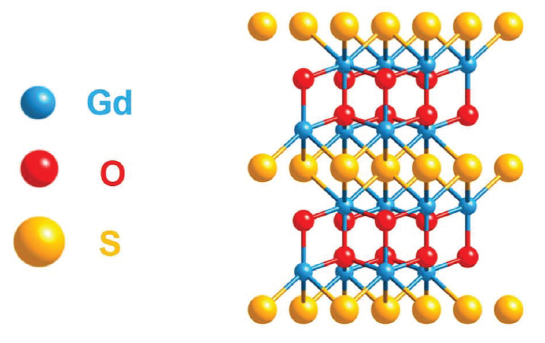
Band Gap Engineering from Cation Balance: The Case of Lanthanide Oxysulfide Nanoparticles
|
C. Larquet, A.-M. Nguyen, E. Glais, L. Paulatto, C. Sassoye, M. Selmane, P. Lecante, C. Maheu, C. Geantet, L. Cardenas, C. Chanéac, A.Gauzzi, C. Sanchez, S. Carenco, Chem. Mater. 2019, 31, 5014-23. |
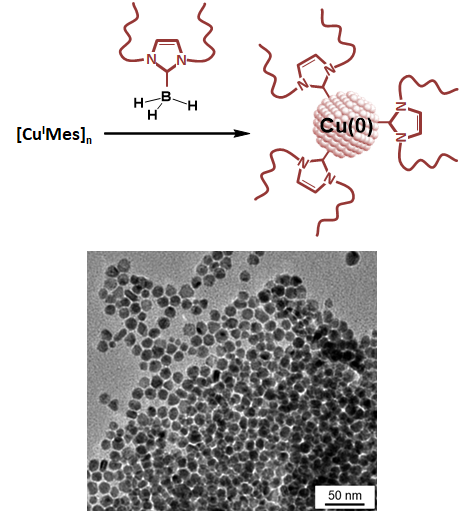
Direct Synthesis of N-Heterocyclic Carbene-Stabilized Copper Nanoparticles from a N-Heterocyclic Carbene-Borane
|
X. Frogneux, L. Hippolyte, D. Mercier, D. Portehault, C. Chaneac, C. Sanchez, P. Marcus, F. Ribot, L. Fensterbank, S. Carenco, Chem. Eur. J. 2019, 25, 11481-85. |
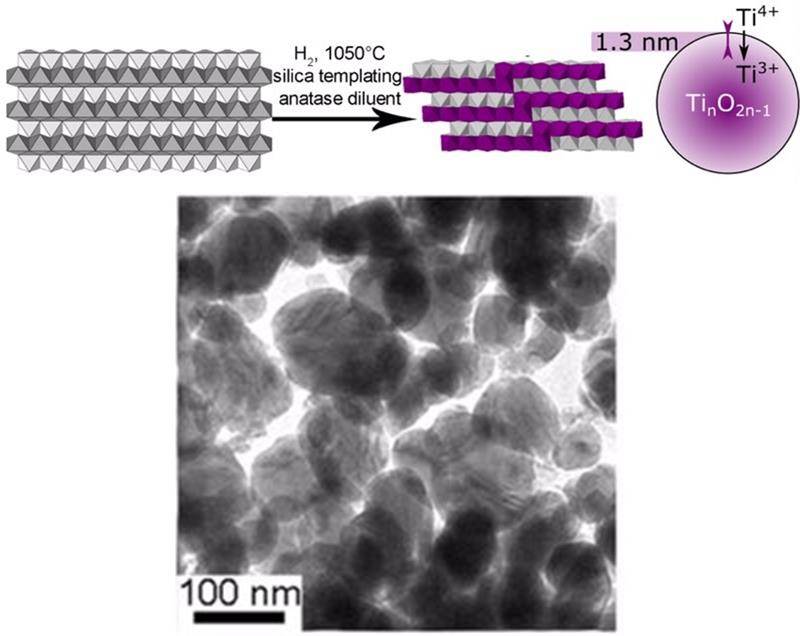
Different reactivity of rutile and anatase TiO2 nanoparticles: synthesis and surface states of nanoparticles of mixed-valence Magnéli oxides
|
E. Baktash, J. Capitolis, L. Tinat, C. Larquet, T. H. C. Chan Chang, J.-J. Gallet, F. Bournel, C. Sanchez, S. Carenco, D. Portehault. Chem. Eur. J. 2019, 25, 11114-20. |
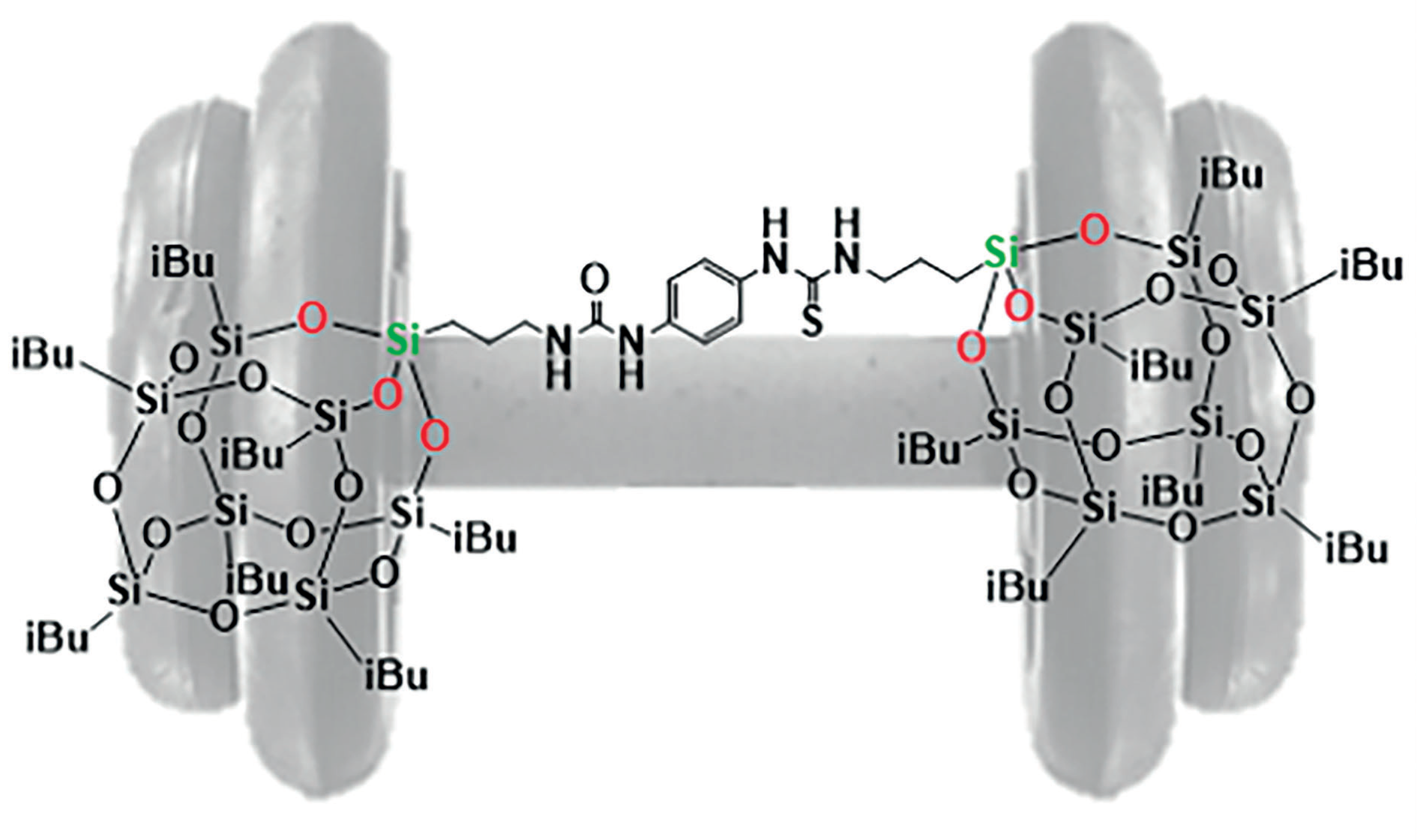
Dumbbell-Shaped T8-POSS with Functional Organic Linkers
|
N. Rey, S. Carenco, C. Carcel, A. Ouali, D. Portehault, M. Wong Chi Man, C. Sanchez, Eur. J. Inorg. Chem. 2019, 27, 3148-56. |
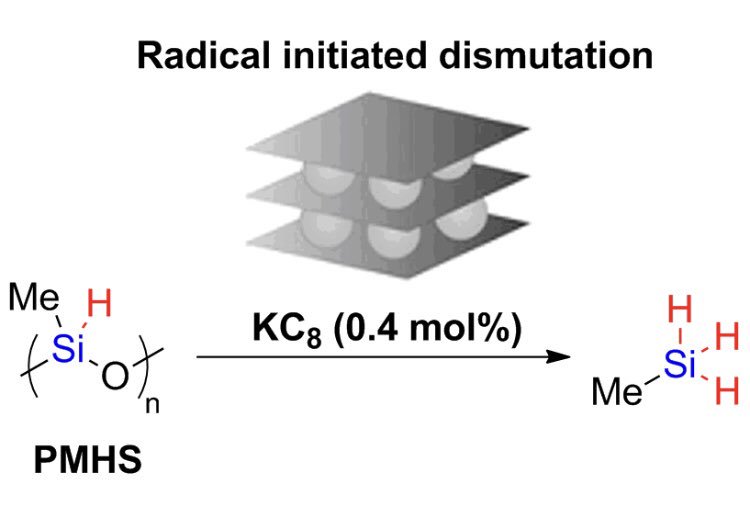
Radical-Initiated Dismutation of Hydrosiloxanes by Catalytic Potassium-Graphite
|
X. Frogneux, A. Pesesse, S. Delacroix, F. Ribot, S. Carenco, ChemCatChem 2019, 11, 3781-85. |
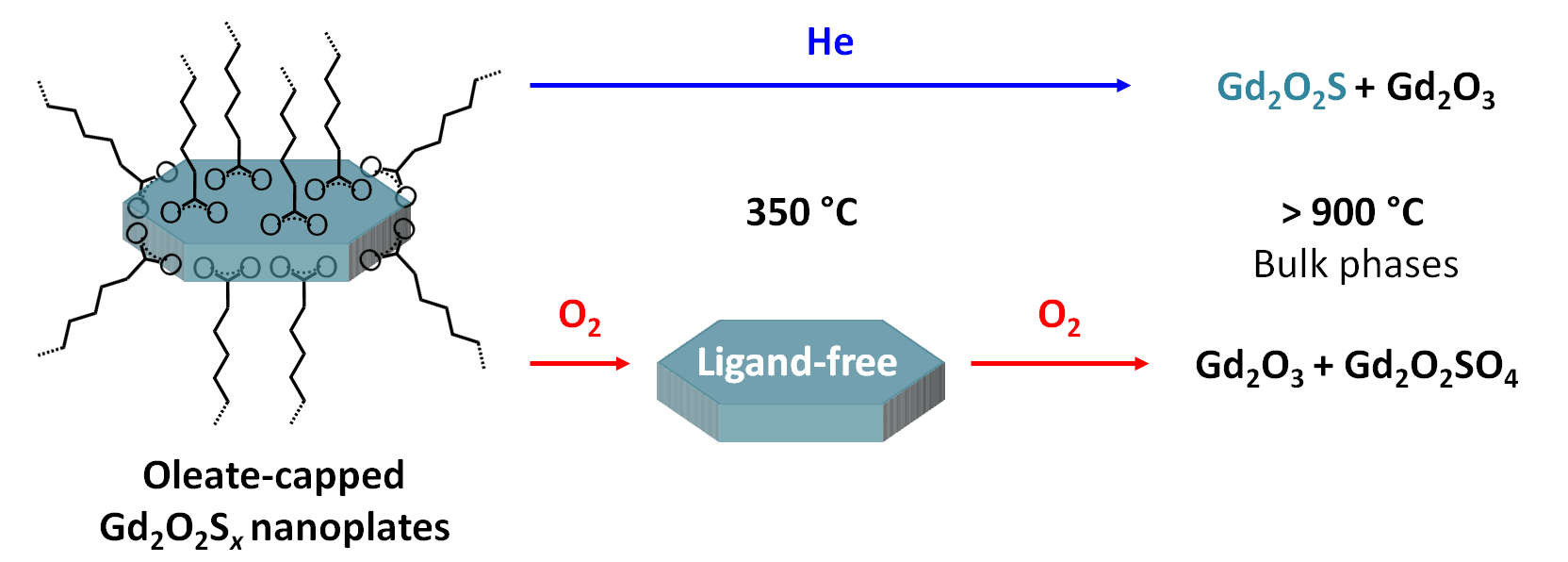
Thermal Stability of Oleate-Stabilized Gd2O2S Nanoplates in Inert and Oxidizing Atmospheres
|
C. Larquet, D. Hourlier, A.-M. Nguyen, A. Torres-Pardo, A. Gauzzi, C. Sanchez, S. Carenco, ChemNanoMat 2019, 5, 539-46. |
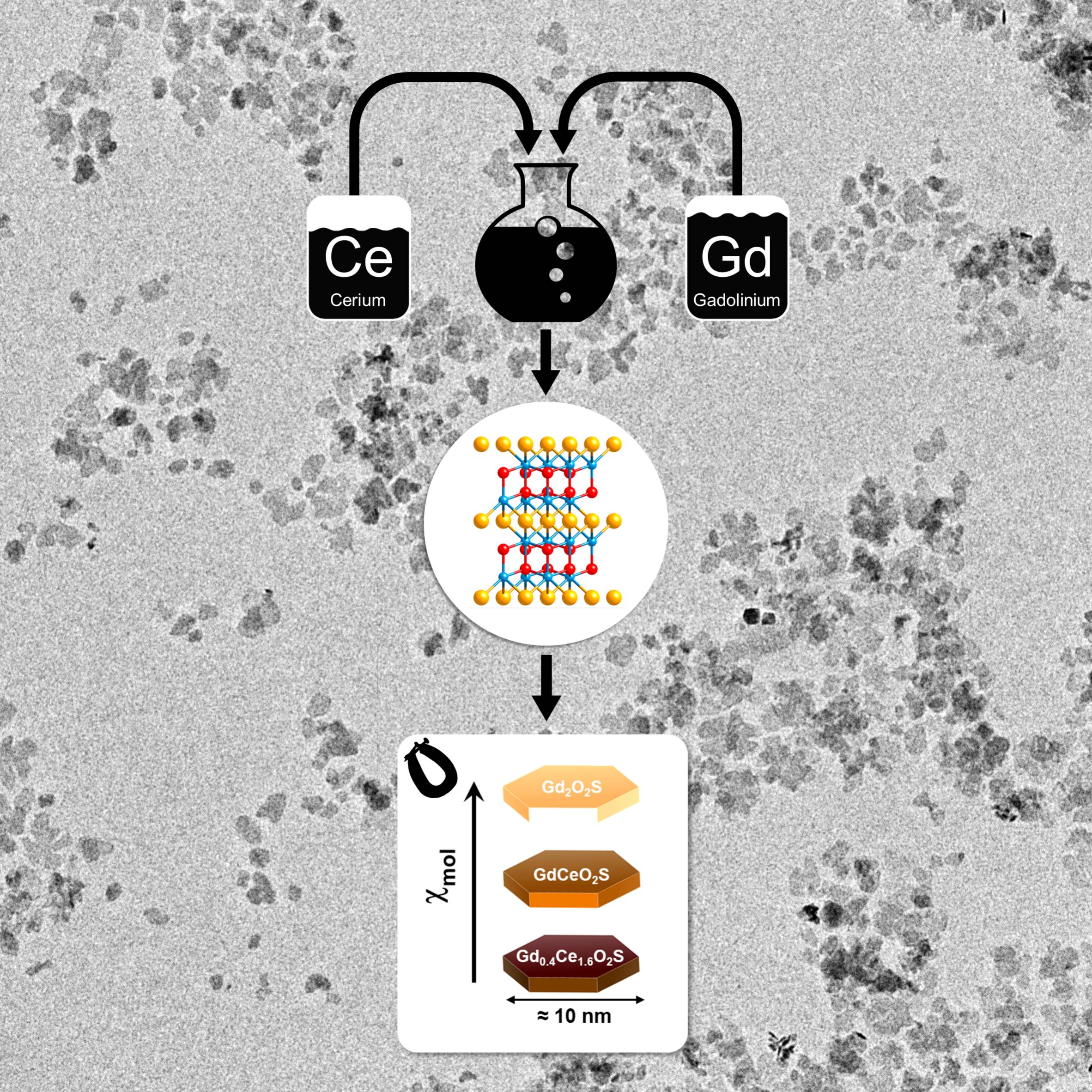
Tunable Magnetic Properties of (Gd,Ce)2O2S Oxysulfide Nanoparticles
|
C. Larquet, Y. Klein, D. Hrabovsky, A. Gauzzi, C. Sanchez, S. Carenco, EurJIc. 2019, 6, 762-765 | EurJIc Cover. |
[Article] Les oxysulfures de lanthanides : Un terrain de jeu pour la nanochimie
|
C. Larquet, A.-M. Nguyen, T. K.-C. Le, M. Avila-Gutierrez, S. Carenco, L'Actualité Chimique. 2019, 436, 28-31. |
[Traduction] Précis de nomenclature en chimie inorganique
|
Ce document est l’adaptation française du "Brief Guide" de l’IUPAC pour la nomenclature en chimie inorganique. Il présente un aperçu des règles essentielles de la nomenclature pour générer noms et formules pour les composés inorganiques, notamment les composés de coordination et les composés organométalliques. |
 |
|
J. Capitolis, S. Delacroix, X. Frogneux, E. Medina, N. Rey, L. Tinat, S. Carenco, L'Actualité Chimique. 2019, 437, 12-18. |