Publications
| 2025 (2) | |||
| 2024 (7) | 2023 (5) | 2022 (3) | 2021 (5) |
| 2020 (3) | 2019 (12) | 2018 (5) | 2017 (3) |
| 2016 (4) | 2015 (2) | 2014 (4) | 2013 (7) |
| 2012 (7) | 2011 (1) | 2010 (2) | 2008 (1) |
2012 | Articles and Reviews
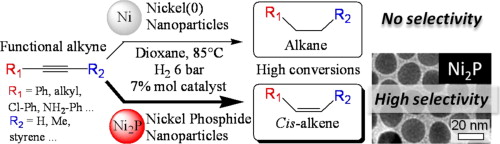
Nickel phosphide nanocatalysts for the chemoselective hydrogenation of alkynes
|
S. Carenco, A. Leyva-Pérez, P. Concepción, C. Boissière, N. Mézailles, C. Sanchez, A. Corma, Nano Today 2012, 7, 21. |
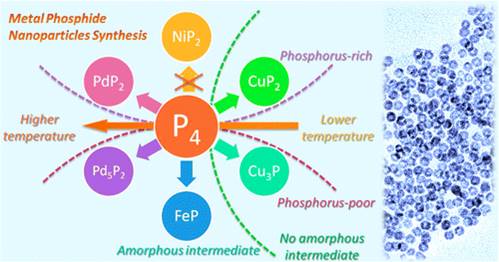
Metal-Dependent Interplay between Crystallization and Phosphorus Diffusion during the Synthesis of Metal Phosphide Nanoparticles
|
S. Carenco, Y. Hu, I. Florea, O. Ersen, C. Boissière, N. Mézailles, C. Sanchez, Chem. Mater. 2012, 24, 4134. |
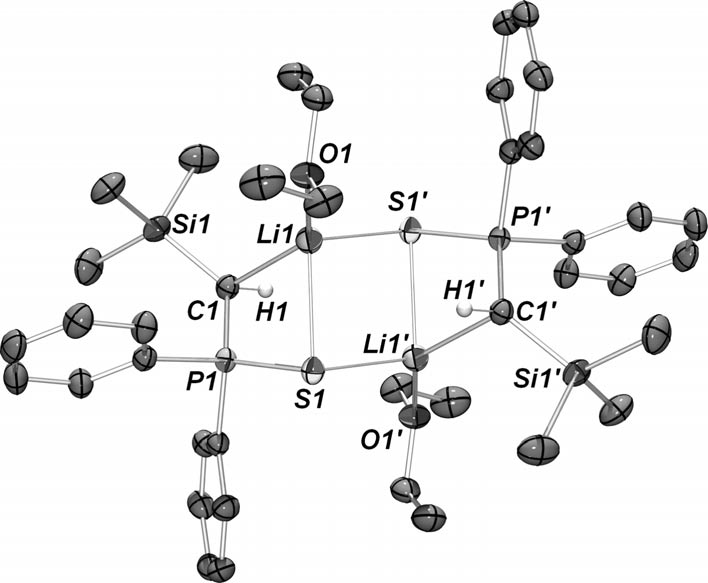
Rhodium (Thiophosphinoyl)(trimethylsilyl)methanide and Bis(thiophosphinoyl)methanide Complexes: S~S vs. C~S Coordination
|
H. Heuclin, S. Carenco, X.-F. Le Goff, N. Mézailles, Eur. J. Inorg. Chem. 2012, 2012, 2320. |
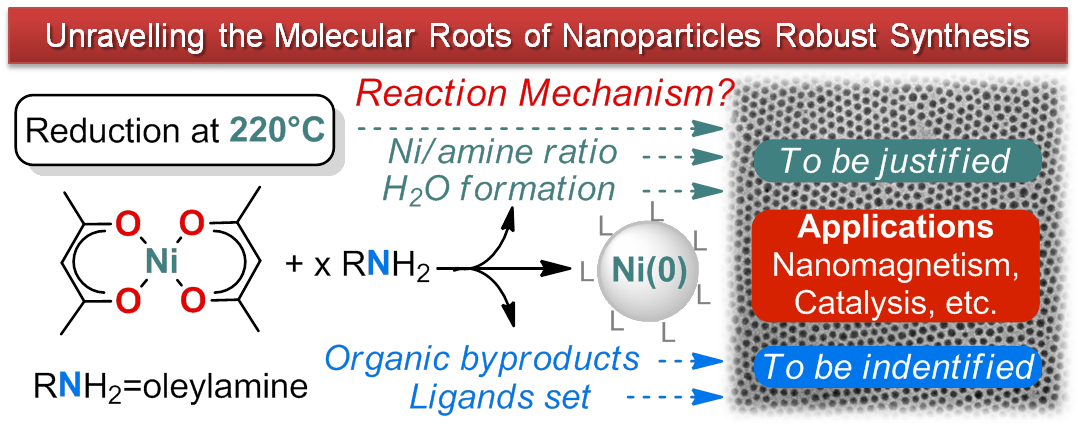
Revisiting the Molecular Roots of a Ubiquitously Successful Synthesis: Nickel(0) Nanoparticles by Reduction of [Ni(acetylacetonate)2]
|
S. Carenco, S. Labouille, S. Bouchonnet, C. Boissière, X.-F. Le Goff, C. Sanchez, N. Mézailles, Chem. - A Eur. J. 2012, 18, 14165. |
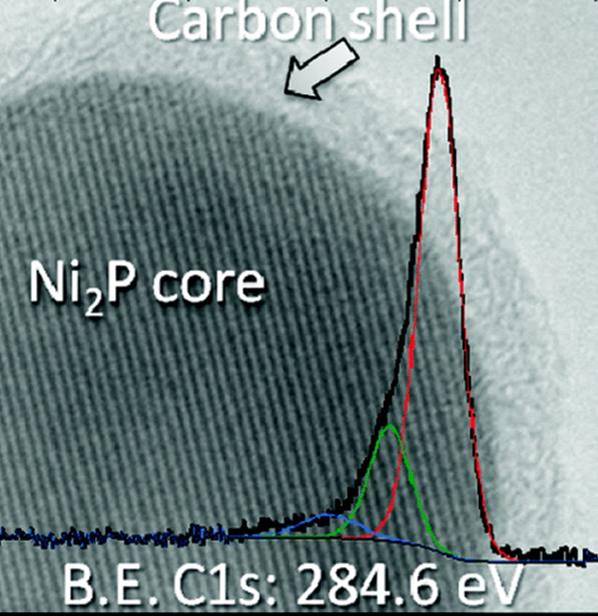
Improving the Li-Electrochemical Properties of Monodisperse Ni2P Nanoparticles by Self-Generated Carbon Coating
|
S. Carenco, C. Surcin, M. Morcrette, D. Larcher, N. Mézailles, C. Boissière, C. Sanchez, Chem. Mater. 2012, 24, 688. |
[Article] Les phosphures de métaux : une renaissance à l'échelle nanométrique
|
S. Carenco, C. Boissière, N. Mézailles, C. Sanchez, Actual. Chim. 2012, 362, 22-28 |
[Book chapter] Metal Phosphides: From Chemist's Oddities to Designed Functional Materials
|
S. Carenco, M. Demange, C. Boissière, C. Sanchez, N. Mézailles, in Mol. Work, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, 2012, pp. 113–120. |